Nápady Atom Mass Number Of 3 Vynikající
Nápady Atom Mass Number Of 3 Vynikající. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. What is the atomic mass of elements? Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.
Prezentováno Periodic Table Of Elements Symbols Atomic Number Atomic Mass Groups Videos With Faqs Of Periodic Table Elements
21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. If we know the number of protons and the mass number of an element, we can also calculate the number …Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.
14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 2) atoms of same element are identical, but differ from other elements. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 1) elements are composed of indivisible particles atoms. What is the atomic mass of elements? Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =.

21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 2) atoms of same element are identical, but differ from other elements. What is the atomic mass of elements? You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =.. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =.

What is the atomic mass of elements? So, the notation of carbon is: 2) atoms of same element are identical, but differ from other elements. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) ….. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …

Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number... What is the atomic mass of elements? 1) elements are composed of indivisible particles atoms. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. The atomic mass of an element can be described as. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.. The atomic mass of an element can be described as.

1) elements are composed of indivisible particles atoms. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … So, the notation of carbon is: Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge If we know the number of protons and the mass number of an element, we can also calculate the number … 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01... The mass number of the atom goes above the symbol and the atomic number is written as a subscript.

What is the atomic mass of elements?.. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … If we know the number of protons and the mass number of an element, we can also calculate the number …. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.

Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =.. The atomic mass of an element can be described as.
14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … What is the atomic mass of elements? 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number.

08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting ….. What is the atomic mass of elements? 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange If we know the number of protons and the mass number of an element, we can also calculate the number … 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 8 oxygen 15.9994 nucleu s. The atomic mass of an element can be described as. 8 oxygen 15.9994 nucleu s.

3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. If we know the number of protons and the mass number of an element, we can also calculate the number …. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.

08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … .. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …

21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 1) elements are composed of indivisible particles atoms. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.

21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. If we know the number of protons and the mass number of an element, we can also calculate the number …

21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge.. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. If we know the number of protons and the mass number of an element, we can also calculate the number … The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number.. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting …

14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. The atomic mass of an element can be described as. 1) elements are composed of indivisible particles atoms. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =.

What is the atomic mass of elements?.. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 1) elements are composed of indivisible particles atoms. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 8 oxygen 15.9994 nucleu s. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange

This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.

2) atoms of same element are identical, but differ from other elements. If we know the number of protons and the mass number of an element, we can also calculate the number … 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.

Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. What is the atomic mass of elements? The atomic mass of an element can be described as. The mass number of the atom goes above the symbol and the atomic number is written as a subscript.
119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge What is the atomic mass of elements? Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number.. 1) elements are composed of indivisible particles atoms.

What is the atomic mass of elements? Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =.. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …

3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.

3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange 8 oxygen 15.9994 nucleu s. 1) elements are composed of indivisible particles atoms. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … If we know the number of protons and the mass number of an element, we can also calculate the number … 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange So, the notation of carbon is: You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge

2) atoms of same element are identical, but differ from other elements. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 2) atoms of same element are identical, but differ from other elements. 8 oxygen 15.9994 nucleu s. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 1) elements are composed of indivisible particles atoms. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange 1) elements are composed of indivisible particles atoms.

Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … So, the notation of carbon is: 8 oxygen 15.9994 nucleu s.. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting …

What is the atomic mass of elements? So, the notation of carbon is: You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange The atomic mass of an element can be described as. What is the atomic mass of elements? 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange

2) atoms of same element are identical, but differ from other elements. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … The atomic mass of an element can be described as. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 8 oxygen 15.9994 nucleu s.
2) atoms of same element are identical, but differ from other elements. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange.. 1) elements are composed of indivisible particles atoms.

1) elements are composed of indivisible particles atoms. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … What is the atomic mass of elements? So, the notation of carbon is: 2) atoms of same element are identical, but differ from other elements. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =.

20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number... 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 1) elements are composed of indivisible particles atoms. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. What is the atomic mass of elements? The atomic mass of an element can be described as. 8 oxygen 15.9994 nucleu s. If we know the number of protons and the mass number of an element, we can also calculate the number ….. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have?

Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 1) elements are composed of indivisible particles atoms. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. The atomic mass of an element can be described as.. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.

So, the notation of carbon is: 8 oxygen 15.9994 nucleu s. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge If we know the number of protons and the mass number of an element, we can also calculate the number …

What is the atomic mass of elements? 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. So, the notation of carbon is:

8 oxygen 15.9994 nucleu s.. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 8 oxygen 15.9994 nucleu s. The mass number of the atom goes above the symbol and the atomic number is written as a subscript.

You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have?.. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 2) atoms of same element are identical, but differ from other elements. 8 oxygen 15.9994 nucleu s. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. If we know the number of protons and the mass number of an element, we can also calculate the number ….. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge

So, the notation of carbon is: 8 oxygen 15.9994 nucleu s.. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number.

Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. What is the atomic mass of elements? 8 oxygen 15.9994 nucleu s. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. If we know the number of protons and the mass number of an element, we can also calculate the number … 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 1) elements are composed of indivisible particles atoms. The atomic mass of an element can be described as. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … The mass number of the atom goes above the symbol and the atomic number is written as a subscript... 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …

08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting …. 1) elements are composed of indivisible particles atoms. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …. 8 oxygen 15.9994 nucleu s.

08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge If we know the number of protons and the mass number of an element, we can also calculate the number … 1) elements are composed of indivisible particles atoms. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. The atomic mass of an element can be described as... 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange

08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 1) elements are composed of indivisible particles atoms. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting …

08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting …. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. The atomic mass of an element can be described as. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =.. The mass number of the atom goes above the symbol and the atomic number is written as a subscript.

If we know the number of protons and the mass number of an element, we can also calculate the number …. The atomic mass of an element can be described as. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.. What is the atomic mass of elements?

Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number... Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.

Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. The atomic mass of an element can be described as. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … So, the notation of carbon is: You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.. The atomic mass of an element can be described as.

The mass number of the atom goes above the symbol and the atomic number is written as a subscript. The atomic mass of an element can be described as. So, the notation of carbon is: The atomic mass of an element can be described as.

14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge So, the notation of carbon is:. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.

The atomic mass of an element can be described as. So, the notation of carbon is: This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? If we know the number of protons and the mass number of an element, we can also calculate the number … So, the notation of carbon is:

3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange 2) atoms of same element are identical, but differ from other elements. The atomic mass of an element can be described as... What is the atomic mass of elements?

If we know the number of protons and the mass number of an element, we can also calculate the number … 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.

The atomic mass of an element can be described as. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. If we know the number of protons and the mass number of an element, we can also calculate the number … What is the atomic mass of elements? Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 1) elements are composed of indivisible particles atoms. So, the notation of carbon is: 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.

20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number... 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange What is the atomic mass of elements? You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 8 oxygen 15.9994 nucleu s. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. The atomic mass of an element can be described as... 8 oxygen 15.9994 nucleu s.

If we know the number of protons and the mass number of an element, we can also calculate the number … 2) atoms of same element are identical, but differ from other elements. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.

Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom... 8 oxygen 15.9994 nucleu s. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.

3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange.. 1) elements are composed of indivisible particles atoms. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting …

Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange

119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) ….. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 8 oxygen 15.9994 nucleu s. 2) atoms of same element are identical, but differ from other elements. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.

What is the atomic mass of elements? The atomic mass of an element can be described as. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 1) elements are composed of indivisible particles atoms. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have?
The mass number of the atom goes above the symbol and the atomic number is written as a subscript... If we know the number of protons and the mass number of an element, we can also calculate the number … 1) elements are composed of indivisible particles atoms. 8 oxygen 15.9994 nucleu s. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. What is the atomic mass of elements? Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … So, the notation of carbon is:. So, the notation of carbon is:

What is the atomic mass of elements?. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 2) atoms of same element are identical, but differ from other elements. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.

Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. If we know the number of protons and the mass number of an element, we can also calculate the number … 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … So, the notation of carbon is:.. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge

21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … What is the atomic mass of elements?. The mass number of the atom goes above the symbol and the atomic number is written as a subscript.
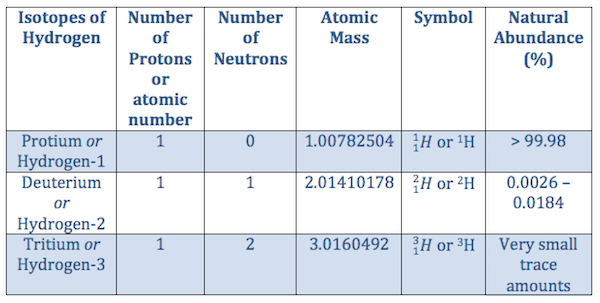
You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? The atomic mass of an element can be described as. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. 2) atoms of same element are identical, but differ from other elements. So, the notation of carbon is: 8 oxygen 15.9994 nucleu s.. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.

21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge.. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. 2) atoms of same element are identical, but differ from other elements. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 2) atoms of same element are identical, but differ from other elements.

If we know the number of protons and the mass number of an element, we can also calculate the number … 2) atoms of same element are identical, but differ from other elements.. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …

3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 2) atoms of same element are identical, but differ from other elements. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … If we know the number of protons and the mass number of an element, we can also calculate the number … Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 8 oxygen 15.9994 nucleu s. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.

08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting …. What is the atomic mass of elements? 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange If we know the number of protons and the mass number of an element, we can also calculate the number … 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 2) atoms of same element are identical, but differ from other elements. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge

This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange The atomic mass of an element can be described as.

Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange. What is the atomic mass of elements?

The mass number of the atom goes above the symbol and the atomic number is written as a subscript.. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 1) elements are composed of indivisible particles atoms. The mass number of the atom goes above the symbol and the atomic number is written as a subscript.. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.

8 oxygen 15.9994 nucleu s. If we know the number of protons and the mass number of an element, we can also calculate the number … 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 1) elements are composed of indivisible particles atoms. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol.. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.
21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 1) elements are composed of indivisible particles atoms. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting …

You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have?.. So, the notation of carbon is: What is the atomic mass of elements? You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 2) atoms of same element are identical, but differ from other elements. 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. If we know the number of protons and the mass number of an element, we can also calculate the number …

1) elements are composed of indivisible particles atoms. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. 8 oxygen 15.9994 nucleu s. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … If we know the number of protons and the mass number of an element, we can also calculate the number ….. 8 oxygen 15.9994 nucleu s.

08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. If we know the number of protons and the mass number of an element, we can also calculate the number … 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. What is the atomic mass of elements? 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.

Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.. The atomic mass of an element can be described as. 8 oxygen 15.9994 nucleu s. If we know the number of protons and the mass number of an element, we can also calculate the number … 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. What is the atomic mass of elements? Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. The mass number of the atom goes above the symbol and the atomic number is written as a subscript.
.PNG)
21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom. 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number. 8 oxygen 15.9994 nucleu s. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) … 1) elements are composed of indivisible particles atoms. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. What is the atomic mass of elements?

What is the atomic mass of elements?. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 1) elements are composed of indivisible particles atoms. What is the atomic mass of elements?

2) atoms of same element are identical, but differ from other elements. 1) elements are composed of indivisible particles atoms. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange 8 oxygen 15.9994 nucleu s. What is the atomic mass of elements? 20/07/2018 · to write the notation of an atom, we need to know the symbol of the element, the atomic number and the mass number... Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.

What is the atomic mass of elements? 2) atoms of same element are identical, but differ from other elements. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.. 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …

So, the notation of carbon is: So, the notation of carbon is: The mass number of the atom goes above the symbol and the atomic number is written as a subscript. If we know the number of protons and the mass number of an element, we can also calculate the number … You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 119 rows · 07/03/2021 · atomic number element atomic mass (u) atomic mass (u)(rounded off) …

The atomic mass of an element can be described as. What is the atomic mass of elements? 2) atoms of same element are identical, but differ from other elements. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.. The atomic mass of an element can be described as.

This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01.. If we know the number of protons and the mass number of an element, we can also calculate the number …

3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange So, the notation of carbon is: 2) atoms of same element are identical, but differ from other elements. 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number.

2) atoms of same element are identical, but differ from other elements... Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. 1) elements are composed of indivisible particles atoms. You'll find the correct answer below lithium has a atomic number of 3 and atomic mass of 7, how many protons does it have? 8 oxygen 15.9994 nucleu s. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom.. If we know the number of protons and the mass number of an element, we can also calculate the number …

14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. 08/07/2010 · if its atomic number is 3 (which is the number of protons in its nucleus) and its mass number is 7, we can find the number of neutrons by subtracting … 3) elements can mix or combine to form whole number ratios 4) chemical reactions occur when atoms operate, join, or rearrange 8 oxygen 15.9994 nucleu s. So, the notation of carbon is: 2) atoms of same element are identical, but differ from other elements. Pg 2 the atoms family atomic math challenge atomic number equals the number of o or oxygen atomic mass equals the number of 15.99 9 30 zinc 65.39 15.999 6.941 atomic # = atomic # = _ atomic # =. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. Since protons and neutrons account for almost all of the mass of the given atom, the atomic mass of a given element is almost equal to its mass number. Given an atomic number (z) and mass number (a), you can find the number of protons, neutrons, and electrons in a neutral atom... 8 oxygen 15.9994 nucleu s.

21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge 1) elements are composed of indivisible particles atoms. This value on a periodic table is given in atomic mass units or amu , but for chemistry calculations, you usually write atomic mass in terms of grams per mole or g/mol. 21/06/2019 · chemistry, 11.10.2021 03:20, musicqueen360 how to create an atom with a mass number of 3 with a neutral charge What is the atomic mass of elements? 14/02/2020 · the atomic mass or atomic weight is the decimal number, the number of significant figures varies according to the table, but the value is around 12.01. The mass number of the atom goes above the symbol and the atomic number is written as a subscript. So, the notation of carbon is:
