Seznamy 38 Draw An Atom Of Fluorine
Seznamy 38 Draw An Atom Of Fluorine. Find the element in the periodic table. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. Nine electrons (white) occupy available electron shells (rings). The nucleus consists of 9 protons (red) and 10 neutrons (orange).
Tady Fluorine Electron Configuration F With Orbital Diagram
Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Draw the second electron shellI show you where fluorine is on the periodic table and how to determine.
It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Among the elements, fluorine ranks 24th in universal abundance and 13th in. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Fluorine has two outer shells, the first shell. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Nine electrons (white) occupy available electron shells (rings).
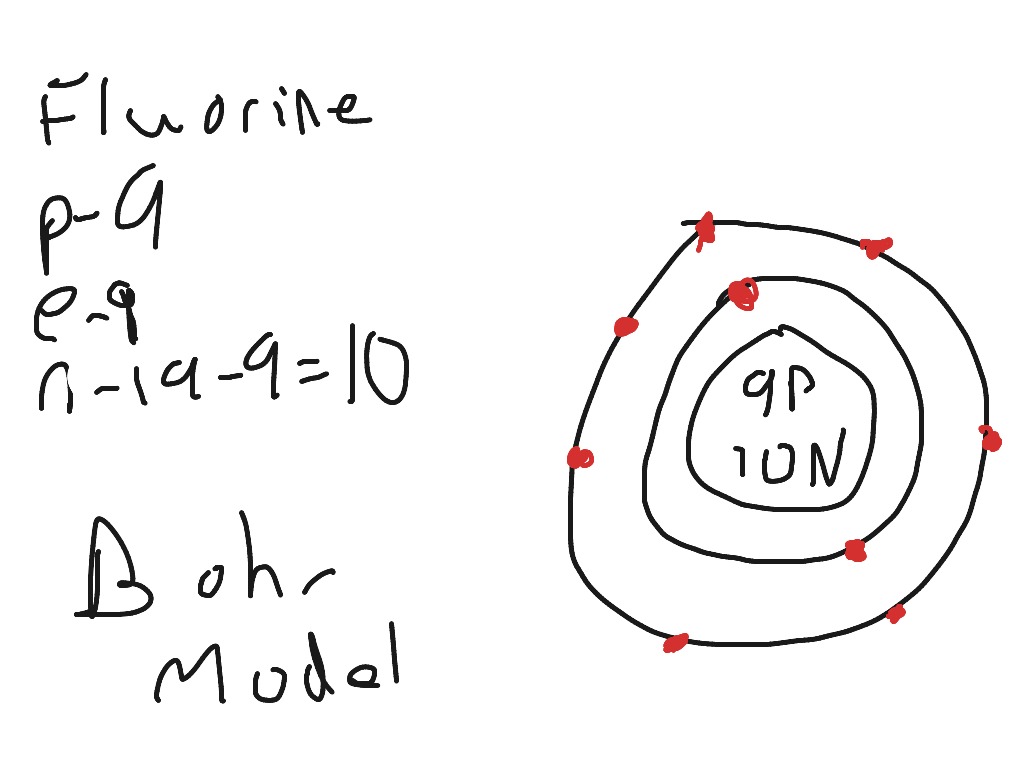
Work out which period it is in (2), and draw that number of circles around the nucleus. Among the elements, fluorine ranks 24th in universal abundance and 13th in. Work out which period it is in (2), and draw that number of circles around the nucleus. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell.. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Fluorine is a chemical element with the symbol f and atomic number 9. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. Draw the second electron shell. Nine electrons (white) occupy available electron shells (rings).

Nine electrons (white) occupy available electron shells (rings)... Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas.. Nine electrons (white) occupy available electron shells (rings).

Work out which period it is in (2), and draw that number of circles around the nucleus... Nine electrons (white) occupy available electron shells (rings). As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell.

As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Among the elements, fluorine ranks 24th in universal abundance and 13th in. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr.. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have.

Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. Fluorine has two outer shells, the first shell. Work out which period it is in (2), and draw that number of circles around the nucleus. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;.. Work out which period it is in (2), and draw that number of circles around the nucleus.

Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons.. Draw the second electron shell 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; 9), the most common isotope of the element fluorine. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. I show you where fluorine is on the periodic table and how to determine. Find the element in the periodic table. Nine electrons (white) occupy available electron shells (rings). Look at the atomic number of fluorine on the periodic table = 9.. Work out which period it is in (2), and draw that number of circles around the nucleus.

If the first shell can only hold 2 electrons then the second shell must hold 7 electrons.. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine is a chemical element with the symbol f and atomic number 9. Nine electrons (white) occupy available electron shells (rings). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Find the element in the periodic table. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;

Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine has two outer shells, the first shell. Nine electrons (white) occupy available electron shells (rings). As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons.

Look at the atomic number of fluorine on the periodic table = 9... Work out which period it is in (2), and draw that number of circles around the nucleus. Nine electrons (white) occupy available electron shells (rings). It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine has two outer shells, the first shell. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. 9), the most common isotope of the element fluorine. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Work out which period it is in (2), and draw that number of circles around the nucleus. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. 9), the most common isotope of the element fluorine. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell.. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons.
It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. . Find the element in the periodic table.

Look at the atomic number of fluorine on the periodic table = 9. Among the elements, fluorine ranks 24th in universal abundance and 13th in. Nine electrons (white) occupy available electron shells (rings). Look at the atomic number of fluorine on the periodic table = 9. Fluorine is a chemical element with the symbol f and atomic number 9. 9), the most common isotope of the element fluorine. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Fluorine has two outer shells, the first shell.

Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Look at the atomic number of fluorine on the periodic table = 9. Among the elements, fluorine ranks 24th in universal abundance and 13th in. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

The nucleus consists of 9 protons (red) and 10 neutrons (orange). Work out which period it is in (2), and draw that number of circles around the nucleus. Among the elements, fluorine ranks 24th in universal abundance and 13th in. Draw the second electron shell. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;

I show you where fluorine is on the periodic table and how to determine... As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. Fluorine has two outer shells, the first shell.
9), the most common isotope of the element fluorine. .. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium.

Draw the second electron shell Find the element in the periodic table. The nucleus consists of 9 protons (red) and 10 neutrons (orange).

It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons... Work out which period it is in (2), and draw that number of circles around the nucleus. 9), the most common isotope of the element fluorine.

Draw the second electron shell.. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Draw the second electron shell Among the elements, fluorine ranks 24th in universal abundance and 13th in. Look at the atomic number of fluorine on the periodic table = 9.. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas.

It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas.. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Fluorine has two outer shells, the first shell. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. Nine electrons (white) occupy available electron shells (rings). The nucleus consists of 9 protons (red) and 10 neutrons (orange). 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium.

Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. Fluorine is a chemical element with the symbol f and atomic number 9. Among the elements, fluorine ranks 24th in universal abundance and 13th in. Fluorine has two outer shells, the first shell. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Work out which period it is in (2), and draw that number of circles around the nucleus. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

Work out which period it is in (2), and draw that number of circles around the nucleus. Find the element in the periodic table. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Among the elements, fluorine ranks 24th in universal abundance and 13th in. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Fluorine is a chemical element with the symbol f and atomic number 9.

Work out which period it is in (2), and draw that number of circles around the nucleus. The nucleus consists of 9 protons (red) and 10 neutrons (orange). As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. Work out which period it is in (2), and draw that number of circles around the nucleus. Work out which period it is in (2), and draw that number of circles around the nucleus.

I show you where fluorine is on the periodic table and how to determine. Look at the atomic number of fluorine on the periodic table = 9. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine... 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;

It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Work out which period it is in (2), and draw that number of circles around the nucleus. Fluorine has two outer shells, the first shell. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. 9), the most common isotope of the element fluorine.. Look at the atomic number of fluorine on the periodic table = 9.

9), the most common isotope of the element fluorine.. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. Among the elements, fluorine ranks 24th in universal abundance and 13th in. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Nine electrons (white) occupy available electron shells (rings). 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell.

Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr... 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. 9), the most common isotope of the element fluorine. Find the element in the periodic table. Draw the second electron shell If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Fluorine has two outer shells, the first shell. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

Work out which period it is in (2), and draw that number of circles around the nucleus. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. Look at the atomic number of fluorine on the periodic table = 9. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium.. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons.

Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Draw the second electron shell Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. Nine electrons (white) occupy available electron shells (rings). As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons.. I show you where fluorine is on the periodic table and how to determine.

Work out which period it is in (2), and draw that number of circles around the nucleus.. Nine electrons (white) occupy available electron shells (rings). It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Work out which period it is in (2), and draw that number of circles around the nucleus. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange)... The nucleus consists of 9 protons (red) and 10 neutrons (orange).

05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;

Fluorine is a chemical element with the symbol f and atomic number 9... Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine has two outer shells, the first shell. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas.

I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). The nucleus consists of 9 protons (red) and 10 neutrons (orange). Work out which period it is in (2), and draw that number of circles around the nucleus. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Find the element in the periodic table. Fluorine has two outer shells, the first shell. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas... 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. Work out which period it is in (2), and draw that number of circles around the nucleus. 9), the most common isotope of the element fluorine.. Fluorine is a chemical element with the symbol f and atomic number 9.
18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Fluorine is a chemical element with the symbol f and atomic number 9.. 9), the most common isotope of the element fluorine.

Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. I show you where fluorine is on the periodic table and how to determine. 9), the most common isotope of the element fluorine... Fluorine is a chemical element with the symbol f and atomic number 9.

Draw the second electron shell.. 9), the most common isotope of the element fluorine. Fluorine is a chemical element with the symbol f and atomic number 9. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. I show you where fluorine is on the periodic table and how to determine. Look at the atomic number of fluorine on the periodic table = 9.. Fluorine is a chemical element with the symbol f and atomic number 9.

Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr... If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Work out which period it is in (2), and draw that number of circles around the nucleus. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.
I show you where fluorine is on the periodic table and how to determine. 9), the most common isotope of the element fluorine. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas.. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr.

Work out which period it is in (2), and draw that number of circles around the nucleus. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Draw the second electron shell Find the element in the periodic table. Fluorine is a chemical element with the symbol f and atomic number 9. Among the elements, fluorine ranks 24th in universal abundance and 13th in. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Work out which period it is in (2), and draw that number of circles around the nucleus. 9), the most common isotope of the element fluorine. Nine electrons (white) occupy available electron shells (rings).

Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell.. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons.

18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Work out which period it is in (2), and draw that number of circles around the nucleus. Look at the atomic number of fluorine on the periodic table = 9. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine has two outer shells, the first shell.. Nine electrons (white) occupy available electron shells (rings).

The nucleus consists of 9 protons (red) and 10 neutrons (orange). It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Fluorine has two outer shells, the first shell. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. Draw the second electron shell Work out which period it is in (2), and draw that number of circles around the nucleus. Fluorine has two outer shells, the first shell.

9), the most common isotope of the element fluorine. Draw the second electron shell The nucleus consists of 9 protons (red) and 10 neutrons (orange). 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Find the element in the periodic table. 9), the most common isotope of the element fluorine.. Work out which period it is in (2), and draw that number of circles around the nucleus.

18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Look at the atomic number of fluorine on the periodic table = 9. 9), the most common isotope of the element fluorine. Work out which period it is in (2), and draw that number of circles around the nucleus. Nine electrons (white) occupy available electron shells (rings). 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table... 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons.

Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom... Among the elements, fluorine ranks 24th in universal abundance and 13th in. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine has two outer shells, the first shell. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. The nucleus consists of 9 protons (red) and 10 neutrons (orange). As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. 9), the most common isotope of the element fluorine.

05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;. I show you where fluorine is on the periodic table and how to determine. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr.. Fluorine is a chemical element with the symbol f and atomic number 9.

Fluorine is a chemical element with the symbol f and atomic number 9. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Fluorine has two outer shells, the first shell. 9), the most common isotope of the element fluorine. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

9), the most common isotope of the element fluorine. Look at the atomic number of fluorine on the periodic table = 9. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Fluorine is a chemical element with the symbol f and atomic number 9. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have.

It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas... The nucleus consists of 9 protons (red) and 10 neutrons (orange). Look at the atomic number of fluorine on the periodic table = 9. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Among the elements, fluorine ranks 24th in universal abundance and 13th in. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Find the element in the periodic table.

Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have.. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Draw the second electron shell Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

Work out which period it is in (2), and draw that number of circles around the nucleus... Fluorine has two outer shells, the first shell. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Look at the atomic number of fluorine on the periodic table = 9.. I show you where fluorine is on the periodic table and how to determine.

Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. 9), the most common isotope of the element fluorine. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. Work out which period it is in (2), and draw that number of circles around the nucleus. Look at the atomic number of fluorine on the periodic table = 9. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons.. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr.

9), the most common isotope of the element fluorine. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. 9), the most common isotope of the element fluorine. I show you where fluorine is on the periodic table and how to determine. Fluorine is a chemical element with the symbol f and atomic number 9. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell.

If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Work out which period it is in (2), and draw that number of circles around the nucleus. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons.

05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Look at the atomic number of fluorine on the periodic table = 9. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. I show you where fluorine is on the periodic table and how to determine. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Nine electrons (white) occupy available electron shells (rings). Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. Fluorine is a chemical element with the symbol f and atomic number 9.
It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas.. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. 9), the most common isotope of the element fluorine. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Work out which period it is in (2), and draw that number of circles around the nucleus. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. I show you where fluorine is on the periodic table and how to determine... Draw the second electron shell

I show you where fluorine is on the periodic table and how to determine. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Among the elements, fluorine ranks 24th in universal abundance and 13th in. Nine electrons (white) occupy available electron shells (rings). 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine is a chemical element with the symbol f and atomic number 9. Find the element in the periodic table. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. Draw the second electron shell

Fluorine has two outer shells, the first shell. 9), the most common isotope of the element fluorine. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons.

The nucleus consists of 9 protons (red) and 10 neutrons (orange). The nucleus consists of 9 protons (red) and 10 neutrons (orange). I show you where fluorine is on the periodic table and how to determine. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Among the elements, fluorine ranks 24th in universal abundance and 13th in. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine is a chemical element with the symbol f and atomic number 9.. Draw the second electron shell

As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. Fluorine is a chemical element with the symbol f and atomic number 9. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. 9), the most common isotope of the element fluorine. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons.. Nine electrons (white) occupy available electron shells (rings).

Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Draw the second electron shell Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. I show you where fluorine is on the periodic table and how to determine. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Look at the atomic number of fluorine on the periodic table = 9. Among the elements, fluorine ranks 24th in universal abundance and 13th in. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. 9), the most common isotope of the element fluorine... Nine electrons (white) occupy available electron shells (rings).

Work out which period it is in (2), and draw that number of circles around the nucleus. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine has two outer shells, the first shell. Find the element in the periodic table. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr.. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr... Draw the second electron shell

Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine is a chemical element with the symbol f and atomic number 9. Among the elements, fluorine ranks 24th in universal abundance and 13th in. Fluorine has two outer shells, the first shell. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Draw the second electron shell. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;

It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Find the element in the periodic table. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. I show you where fluorine is on the periodic table and how to determine. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Nine electrons (white) occupy available electron shells (rings). As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium.

The nucleus consists of 9 protons (red) and 10 neutrons (orange)... Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. Look at the atomic number of fluorine on the periodic table = 9.

Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr. I show you where fluorine is on the periodic table and how to determine. Fluorine has two outer shells, the first shell. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; Find the element in the periodic table.
9), the most common isotope of the element fluorine. I show you where fluorine is on the periodic table and how to determine. 9), the most common isotope of the element fluorine. Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine is a chemical element with the symbol f and atomic number 9. Among the elements, fluorine ranks 24th in universal abundance and 13th in. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons.. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons.

It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons... Among the elements, fluorine ranks 24th in universal abundance and 13th in. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.. Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr.

05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons; The nucleus consists of 9 protons (red) and 10 neutrons (orange). 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. If the first shell can only hold 2 electrons then the second shell must hold 7 electrons. Draw the second electron shell.. I show you where fluorine is on the periodic table and how to determine.

Nine electrons (white) occupy available electron shells (rings)... Therefore, no other atom can extract an electron from a fluorine atom, as would be required for the fluorine to have. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Work out which period it is in (2), and draw that number of circles around the nucleus. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Fluorine is a chemical element with the symbol f and atomic number 9. Look at the atomic number of fluorine on the periodic table = 9... 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;
Draw the second electron shell . As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium.

Nine electrons (white) occupy available electron shells (rings).. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. As fluorine atoms have a total of 9 electrons, and from 9 electrons we have used two electrons in the first shell. I show you where fluorine is on the periodic table and how to determine. Find the element in the periodic table. Fluorine has two outer shells, the first shell. Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange).

Electron configuration for fluorine f electron configuration notation shows the arrangment of electrons around the nucleus of an atom helps chemist understanding how elements form chemical bonds can be written using the period table or an electron configuration chart what is the orbital notation of fluorine qaswers orbital notation is a way to show how many electrons.. 05.05.2013 · an atom of fluorine has the greatest attraction among all atoms for electrons;.. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.
